The Need to Subtype Type 2 Diabetes: Why and Why not?
By: Gabriel V. Jasul, Jr., M.D., FPCP, FPCEDM
Type 2 diabetes is a metabolic disorder characterized by complex and heterogenous factors and is mainly diagnosed based on the measurement of a single metabolite, blood glucose. The natural history of the development of hyperglycemia is however dependent on those many factors, both genetic and environmental. Consequently, the clinical presentation, response to treatment, development of complications and long-term prognosis invariably differ from one patient to another. Clearly, better understanding of the pathogenetic mechanisms leading to the development of type 2 diabetes has evolved and has improved treatment with newer and better drugs and more effective management strategies. However, the ability to stratify patients into groups directed to the best individualized treatment continues to be challenging.
A recent publication on the cluster analysis of patients with newly diagnosed diabetes from the Swedish All New Diabetics in Scania (SANDIS) cohort (n=8980) by Ahlqvist, et al addressed this challenge. Using six variables that were easily measured in a single clinic visit and reasonably unaffected by treatment, the authors were able to identify five replicable clusters of patients representing five subtypes with significantly different characteristics and different risk of complications. The six variables used were the following: age at diagnosis, body mass index (BMI), glutamate decarboxylase antibodies (GADA) (to identify patients with autoimmune diabetes), HbA1c (to assess blood glucose control), homeostatic model assessment 2-B (HOMA 2-B) (to assess Beta cell function based on C-peptide concentration) and HOMA 2-IR ( to assess insulin sensitivity).
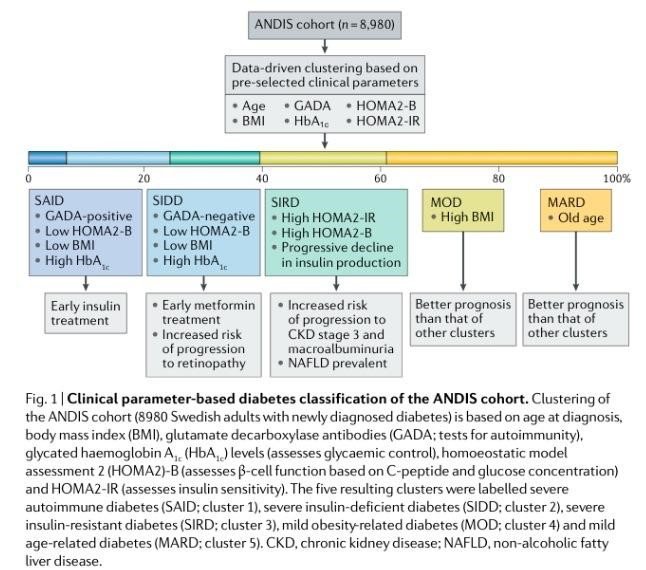
Figure 1. Clinical parameter-based diabetes classification of the ANDIS cohort. (From Rossing, P. Nature Reviews Nephrology 2018)
Figure 1 (from Rossing, P., Nature Reviews Nephrology 2018) summarizes the five identified clusters of patients from the SANDIS cohort. Cluster 1 patients (6.4% of the cohort), labelled severe autoimmune diabetes (SAID), had low BMI, high HbA1c levels, low HOMA-2B index and were GADA-positive. Cluster 2 patients (17.5% of the cohort), labelled severe insulin-deficient diabetes (SIDD), had low BMI, high HbA1c levels, lowest HOMA-2B index and were GADA-negative. Cluster 3 patients (15.3 % of the cohort), labelled severe insulin-resistant diabetes (SIRD) had the highest HOMA-2B IR and HOMA 2-B indices. Cluster 4 patients (21.6% of the cohort), labelled mild obesity-related diabetes (MOD) had the highest BMI and had relatively good prognosis. Cluster 5 patients (39.1% of the cohort), labelled mild age-related diabetes (MARD), had older-age onset and good prognosis.
This clustering was replicated in three other independent cohorts, the Scania Diabetes Registry (n=1466), All New Diabetics in Uppsala (n=844), and Diabetes Registry Vaasa (n=3485), bringing the total number of patients evaluated to 14,755. Important patterns and similar clinical trajectories were observed from the original cluster analysis as well as in the replication analysis. Despite the difference in their GADA status, SAID and SIDD (cluster 1 and 2) patients both had low BMI, insulin deficiency, high HbA1c concentration upon diagnosis, and poorer control when treated. On the other hand, cluster 4 and 5 patients had relatively milder and better-controlled diabetes than SAID or SIDD patients, developed kidney and vascular complications at a slower rate, with MOD (cluster 4) patients being obese and MARD (cluster 5) patients being older at onset.
SIRD (cluster 3) patients, as characterized, had higher HOMA2-B and HOMA2-IR scores reflecting incomplete Beta-cell compensation for insulin resistance and decreasing C-peptide concentrations over 8 years of follow-up. SIRD patients had better control with treatment and yet, were more likely to develop end-stage kidney disease and cardiac events. The increased incidence of diabetic kidney disease (DKD) inspite of relatively lower HbA1c suggests that glucose lowering is not the optimum preventive measure in DKD, especially in SIRD patients. Retinopathy, on the other hand, was more observed in the insulin-deficient clusters, especially in SIDD patients, implying that insulin deficiency or hyperglycemia are important triggers for retinopathy.
Interestingly, a retrospective cohort study from the Steno Diabetes Center Copenhagen (SDCC) (n=2,290) showed that patients with type 2 diabetes clustered into five similar clinically relevant sub-groups. The clinical measures used in the SDCC patients were age at onset, diabetes duration, HbA1c, BMI, HOMA2-B, HOMA2-IR, and GAD65 autoantibodies. The resulting sub-groups were primarily characterized by autoimmune Beta-cell failure (3%), insulin resistance with short disease duration (21%), non-autoimmune Beta-cell failure (22%), insulin resistance with long disease duration (32%), and presence of metabolic syndrome (22%). These sub-groups resemble that of the subtypes observed in the study by Ahlqvist. Retinopathy was also more common in the non-autoimmune Beta-cell failure (corresponding to SIDD), 52% vs 22% in the other sub-groups. Nephropathy was more common in the metabolic syndrome sub-group and cardiovascular disease (CVD) was more common in the sub-group with insulin resistance and long diabetes duration. The prevalence of cardiovascular disease, nephropathy and neuropathy, although different between the SDCC sub-groups, lost statistical significance after adjustment.
Why then it becomes important and critical to stratify and subtype patients with type 2 diabetes? The recent studies on subgrouping type 2 diabetes illustrate that more precise characterization of type 2 diabetes will help to fully inform clinicians and patients on the most appropriate choice of therapeutic agents. Together with relevant genotyping, the possibility of individualizing treatment choices should lead to precision medicine in the truest sense. Knowing the higher risk of developing specific diabetic complications in specific sub-groups will direct clinicians to treat early and aggressively those patients in the high-risk groups with specific drugs that would be more specific and advantageous, e.g., cardioprotective, renoprotective, weight-reducing or more. Glycemic control appears to be not the only major contributory factor in DKD and CVD and therefore addressing the non-glycemic factors aggressively in high-risk subgroups is warranted.
The next question is why would it be difficult and problematic to proceed with subtyping of type 2 diabetes. It is clear that these studies involve essentially homogeneous Scandinavian populations and therefore their results can not be generalized to other populations, especially those residing in so-called diabetes hotspots, especially the Asia-Pacific and Middle East countries. Specific issues, aside from racial/ethnic differences, also have to be considered, including length of follow-up, treatment given to the groups, level of glycemic control and management of blood pressure, lipid and weight during the study period. While the markers used in the studies were considered easily done in the clinical setting in Scandinavia, the tests are not universally available and may not be cheap in the usual practice environment in our country.
Despite these current limitations, subtyping and subgrouping type 2 diabetes would lead to improved patient care and outcomes. Initiatives to perform clinical studies involving Filipinos with diabetes should be our priority and should be undertaken without delay. Moving towards individualized treatment, a real form of precision medicine, in diabetes care is certainly logical. However, at this time, the current practice environment, particularly in the Philippine setting, precludes actual implementation of such strategy. In the future, hopefully sooner than later, local evidence base and resources will make precision medicine feasible and accessible for the Filipino patients with diabetes.
References:
1. Ahlqvist, E. et al. Novel subgroups of adult-onset diabetes and their association with outcomes: a data-driven cluster analysis of six variables. Lancet Diabetes and Endocrinology. Published online March 1, 2018.
2. Sladek, R. The many faces of diabetes: Addressing the heterogeneity of a complex disorder. Lancet Diabetes and Endocrinology. Published online March 1, 2018.
3. Rossing, P. Subclassification of diabetes based on quantitative traits. Nature Reviews Nephrology 2018.
4. Chatterjee, S. and Davies, MJ. Accurate diagnosis of diabetes mellitus and new paradigms of classification. Nature Reviews Endocrinology 2018.
5. Saifai, N., Ali, A., Rossing, P., and Ridderstrale, M. Stratification of type 2 diabetes based on routine clinical markers. Diabetes Research and Clinical Practice. Accepted manuscript, May 8, 2018.
About the author:
Dr. Gabriel V. Jasul, Jr. is currently Clinical Associate Professor at the University of the Philippines College of Medicine-Philippine General Hospital and is concurrent Head of the Diabetes, Thyroid and Endocrine Center and Chief of the Section of Endocrinology, Diabetes and Metabolism at the St. Luke’s Medical Center Quezon City. Dr. Jasul is Past President of the Philippine College of Endocrinology, Diabetes and Metabolism (PCEDM) (2011) and of the Philippine Association for the Study of Overweight and Obesity (PASOO) (2012-2014). He served as Member of the Philippine Specialty Board of Endocrinology, Diabetes and Metabolism (2016-2018). He sits in the Editorial Board of the Journal of the ASEAN Federation of Endocrine Societies (JAFES) as Associate Editor.