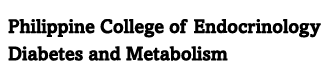Maria Nikki Cruz, MD
The Asean Federation of Endocrine Societies (AFES) returns face-to-face with the 22nd AFES Congress held at Queen Sirikit Convention Center, Bangkok, Thailand last November 16-19, 2023. Speakers from different international and Asean countries convened to present latest updates in the field of Endocrinology.
 Photo: Thaiendocrine Thailand
Photo: Thaiendocrine Thailand
Personalized Treatment in Acromegaly
Assoc. Prof. Sebastian Niggers (Netherland)
Surgery remains the first line treatment for acromegaly. Tumor extension, based on the Knosp criteria, and biochemical activity, preoperative GH and IGF1, remain as the best predictors of surgical remission.
Somatostatin receptor ligands (SRLs) are considered when there is persistence of disease after surgery. The principal molecular determinant predicting response to SRL is SST2 expression. In addition to immunochemistry staining, hypointensity on T2-weighted imaging predicts response to SRL.
An alternative recommendation from Dr. Niggers was the combination of Cabergoline with SRLs if disease is not controlled with SRL monotherapy, emphasizing that the best predictor of remission with Cabergoline use is IGF1 at baseline and after surgery and not prolactin co-secretion.
Furthermore, he recommended that if the disease is not controlled on SRL, clinicians should consider surgery rather than increasing the dose of SRL. Various studies have shown that higher doses have a low percentage of biochemical control. There was no significant difference between high dose and high frequency dosing. An alternative recommendation is the combination of SRL with Pegvisomant which offers a lower cost with good efficacy rate since the dose of SRL can be lowered with pegvisomant.
What is the role of Eucaloric Very-Low-Carbohydrate Ketogenic Diet in Acromegaly Treatment? A eucaloric very-low-carbohydrate ketogenic diet (<50 g of carbohydrate per day) induces ketosis and reduces portal insulin concentrations, which then down-regulate hepatic growth hormone receptors and reduce IGF-I synthesis. A pilot study done in a small population showed the ability of an adjuvant eucaloric very-low-carbohydrate ketogenic diet to result in IGF-I control in patients with acromegaly whose disease was uncontrolled with first-generation somatostatin receptor ligand therapy.
Source: N Engl J Med 2020; 382:2161-2162 DOI: 10.1056/NEJMc1915808
Subclinical Cushing’s Syndrome
Prof. Bien J. Matawaran (Philippines)
Subclinical Cushing’s Syndrome (sCS) also called mild hypercortisolism is a common finding in patients with adrenal incidentaloma. It was defined as the absence of signs and symptoms of cortisol excess, ACTH dependent on biochemical testing and with associated adverse effects of hypercortisolism such as bone fragility, diabetes and hypertension.
Etiologies include 11B-HSD enzyme shuttle, genomic causes, medical disorders associated with chronically activated HPA axis such as renal failure and uncontrolled DM (functional hypercortisolism), circadian dysregulation, stress or chronic stress early in life and exposure to Bisphenol A (BPA).
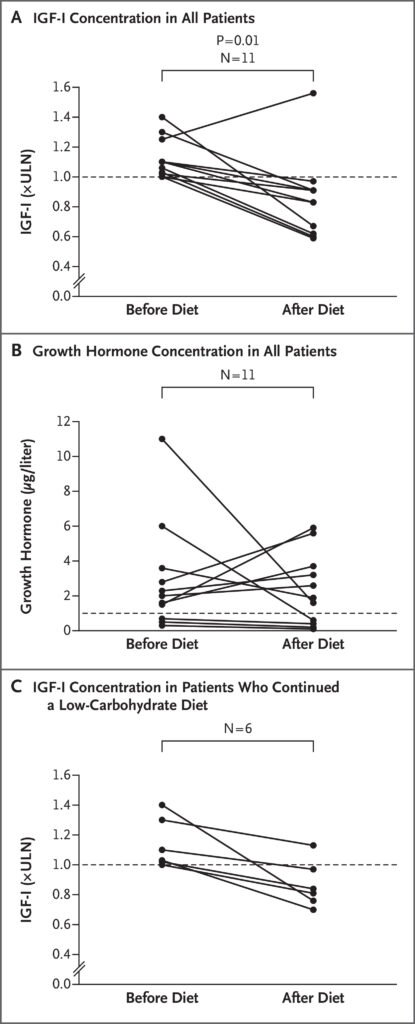
Dr. Matawaran presented the new diagnostic criteria for adrenal sCS by the Japan Endocrine Society.
Source: https://doi.org/10.1507/endocrj.EJ17-0456
Furthermore, the French Society of Endocrinology, Italian Association of Clinical Endocrinologists and European Society of Endocrinology/European Network for the Study of Adrenal Tumors recommended the use of urine free cortisol using UFC-ACTH-DST criteria to support the diagnosis and to identify patients that would need adrenalectomy. The criteria included the following: (1) serum cortisol >3ug/dL after a 1-mg DST, (2) UFC of >70ug/24H and (3) basal ACTH of <10pg/mL. Fulfillment of 2 of the 3 criteria warrants adrenalectomy.
In the Japan Endocrine Society, surgery is recommended for patients with serum cortisol ≥5 μg/dL after a 1-mg DST, especially if treatment-resistant clinical problems such as hypertension, systemic obesity, impaired glucose tolerance, osteoporosis or dyslipidemia are present. At present, only Metyrapone, Ketoconazole and Mifepristone are proven to have an effect on sCS.
Challenging Clinical Cases in testosterone Replacement Therapy in Men
Prof. Vin Tangpricha (USA)
How do we treat Idiopathic Hypothalamic Hypogonadism?
Functional hypogonadotropic hypogonadism (FHH) is defined as the coexistence of androgen deficiency-like features and low serum testosterone concentrations, occurring in the absence of both intrinsic structural hypothalamic-pituitary-gonad (HPG) axis pathology and of specific pathologic conditions suppressing the HPG axis.
Testosterone replacement therapy is the usual first-choice therapy however, it produces a negative feedback on the HPG axis, resulting in suppression of intratesticular testosterone production and spermatogenesis. Therefore, in hypogonadal men of an unidentified cause seeking fertility, clomiphene citrate has been shown to be effective in raising serum testosterone within 6 weeks. It can be started at 25mg or 50mg every other day. FSH and LH were not predictors of response. Long term studies done >5 years showed a sustained increase in testosterone.
Is it safe to continue testosterone replacement therapy in middle-aged and older men with hypogonadism?
The newly published TRAVERSE STUDY is a multicenter, randomized, double-blind, placebo-controlled, noninferiority trial of 5246 men 45 to 80 years of age who had preexisting or a high risk of cardiovascular disease and who reported symptoms of hypogonadism and had two fasting testosterone levels of less than 300 ng per deciliter. Patients were randomly assigned to receive daily transdermal 1.62% testosterone gel (dose adjusted to maintain testosterone levels between 350 and 750 ng per deciliter) or placebo gel. The primary cardiovascular safety endpoint was the first occurrence of any component of a composite of death from cardiovascular causes, nonfatal myocardial infarc- tion, or nonfatal stroke, assessed in a time-to-event analysis. Results showed that testosterone-replacement therapy was noninferior to placebo with respect to the incidence of major adverse cardiac events.

Source: N Engl J Med 2023;389:107-17.DOI: 10.1056/NEJMoa2215025
Adrenal Incidentaloma 2023
Prof. Paul Michael Stewart (United Kingdom)
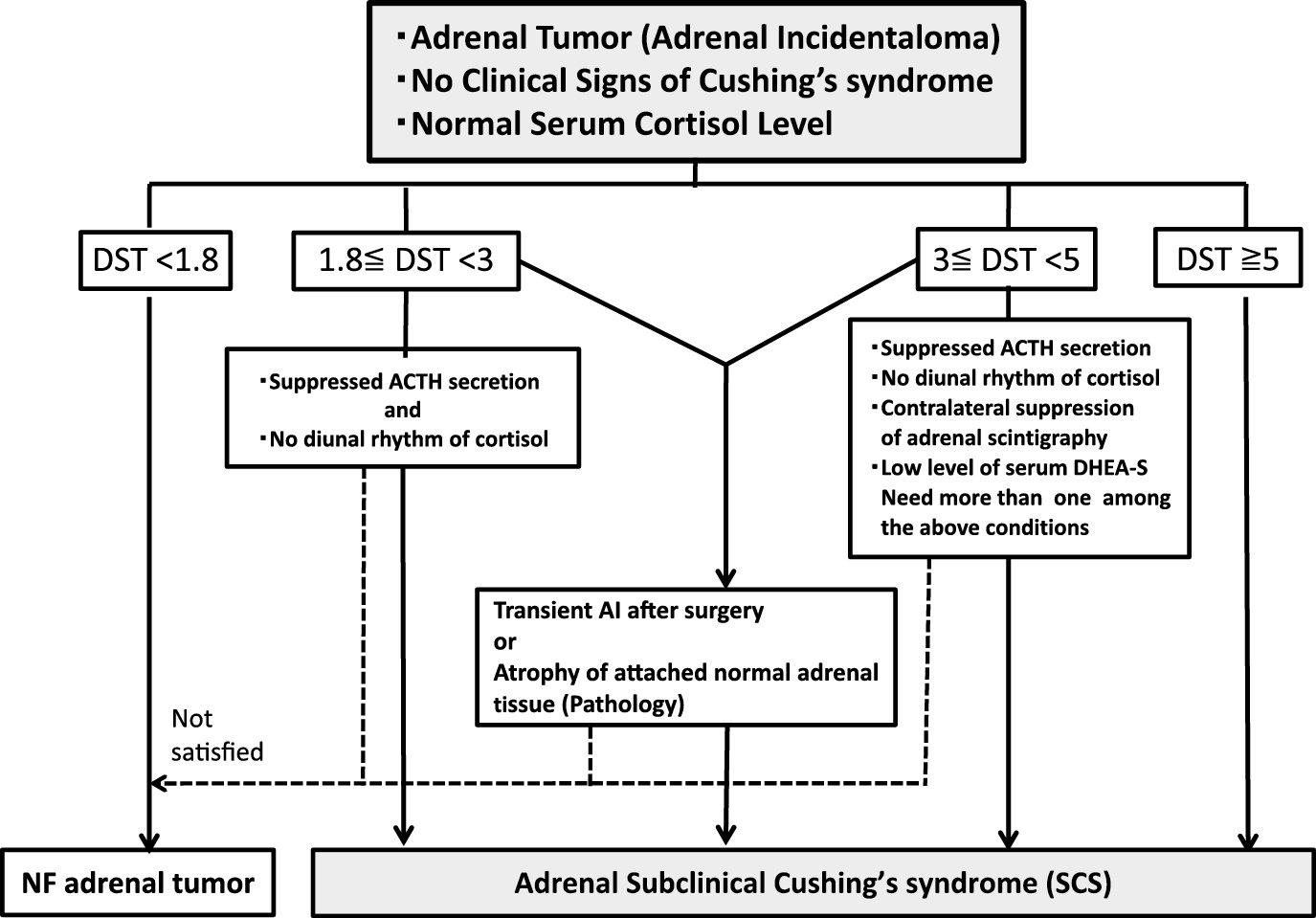
Dr. Steward discussed the 2023 Guidelines on Adrenal Incidentaloma. Differentiation between a benign or malignant nodule should be prioritized based on unenhanced CT features of the nodules. Adrenal biopsy is almost never done unless all of the following criteria are present: (1) The lesion is not conclusively benign on imaging criteria (2) The lesion is hormonally inactive with pheochromocytoma excluded and (3) Outcome will affect therapeutic strategy.
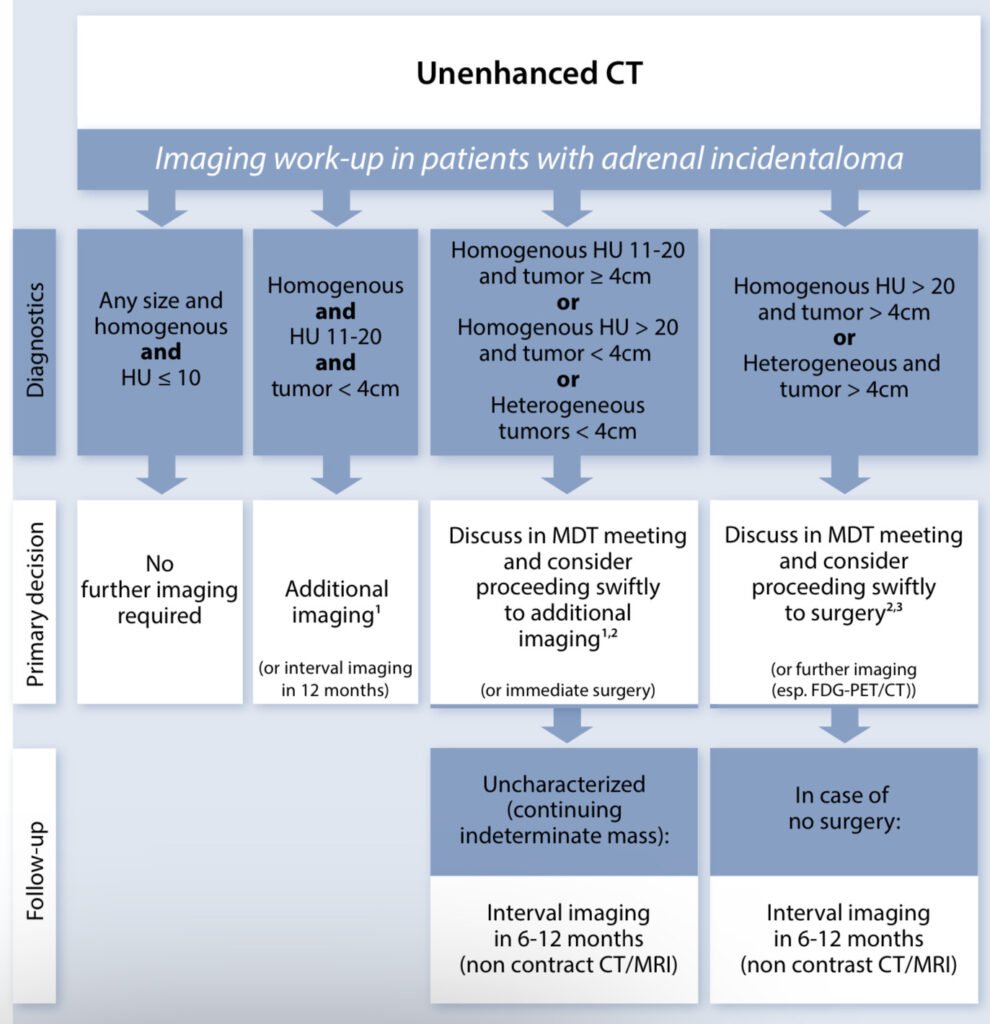
Source: https://doi.org/10.1093/ejendo/lvad066
Patients with lesions non-typical for benign adenoma should have 24H urinary fractionated metanephrines or preferably plasma free metanephrines. DHEAS, 17-OHP, androstenedione and assessment for adrenal insufficiency should be done for nodules with features of malignancy, bilateral nodules/masses and if the patient presents with hyperandrogenism. Plasma renin activity and aldosterone should be done if patients present with hypertension or hypokalemia. For patients with a history of extraadrenal malignancy, pheochromocytoma should still be excluded.
A topic of debate was on cortisol hypersecretion. It was found that mild autonomous cortisol secretion (MACS) is the most common hormonal abnormality found in benign adrenocortical tumors using 1mg Dexamethasone suppression test (DST). However, it was emphasized that pitfalls such as the lack of normative data across population, age and age-related comorbidities (hypertension, glucose intolerance, obesity and osteoporosis) being confounders increase false positive rate in DST. Dr. Stewart emphasized the need to differentiate autonomy versus physiological hypercortisolism. It should be proven that it is autonomous before considering adrenal surgery.
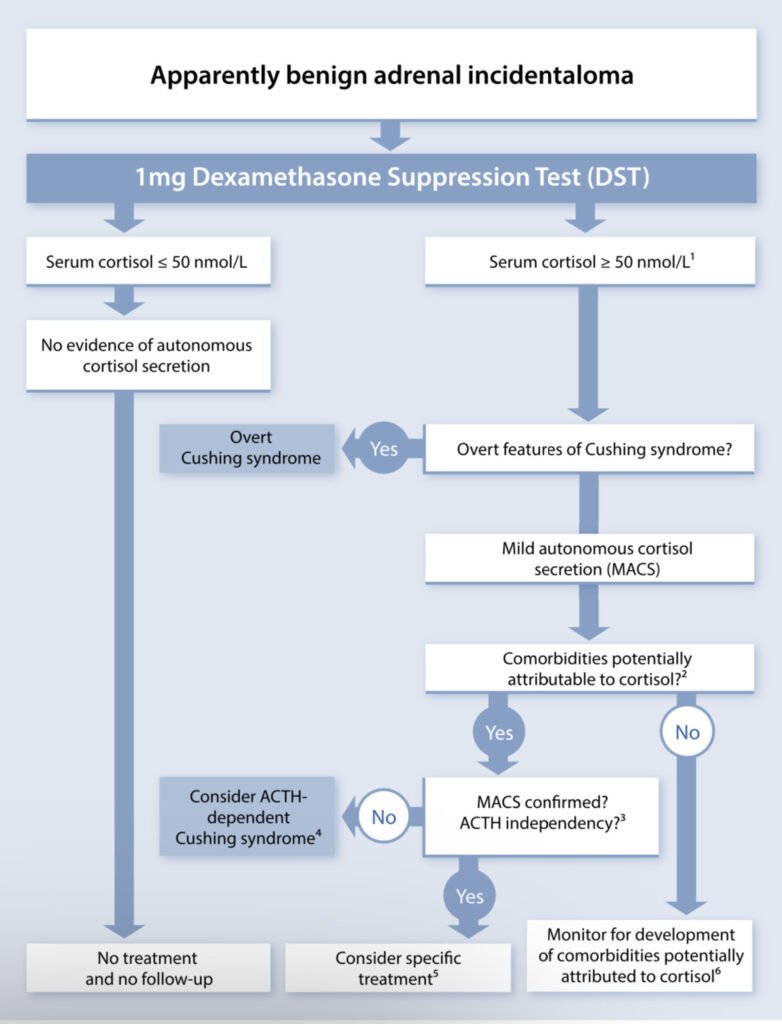
Source: https://doi.org/10.1093/ejendo/lvad066
When adrenocortical carcinoma is suspected by imaging or clinical features, guidelines suggest measurement of sex steroids and precursors of steroidogenesis using multi-steroid profiling. Patients with malignant lesions will have high urinary androgen and glucocorticoid precursors
Optimizing Glucocorticoid Replacement Therapy
Prof. Paul Michael Stewart (United Kingdom)
A common cause of adrenal insufficiency is glucocorticoid suppression from synthetic glucocorticoids. The magnitude of its effect depends on the dose, potency of steroid, half life, duration and mode of administration and concomitant medications. Early recognition is necessary due to the associated increased mortality from synthetic GC use, decreased QoL, decreased bone mass and increased incidence of diabetes, depression, anxiety, hyperlipidemia and hypertension. Diagnosis of GC-induced AI can be done using random cortisol and short synacthen.
One important point raised during the discussion was on steroid overtreatment. Steroidomics showed that physiologic dosing of hydrocortisone was associated with increased THF+allo-THF/THE with no change in EFF/UFE. Immediate release formulations were not shown to provide circadian replacement. With these findings, new medications have been developed in pursuit of circadian therapy. Plendaren is a dual-release formulation of HC that has been used for the treatment of patients with AI that has been associated with reduction of glucocorticoid exposure and reduction in body weight, BP and improvement in glucose tolerance (DREAM study). Efmody is another modified-release hard capsule hydrocortisone licensed in Europe last 2021 for the treatment of patients with CAH over 12 years of age. Lastly, prednisolone is another corticosteroid with a half-life of 3 hours with the same pharmacokinetics as Plenadren.
Lastly, Dr. Stewart emphasized to never suddenly stop steroids without an assessment of GC-AI assessment. The figure below shows the risk assessment from the study of Prete published in BMJ last 2021.
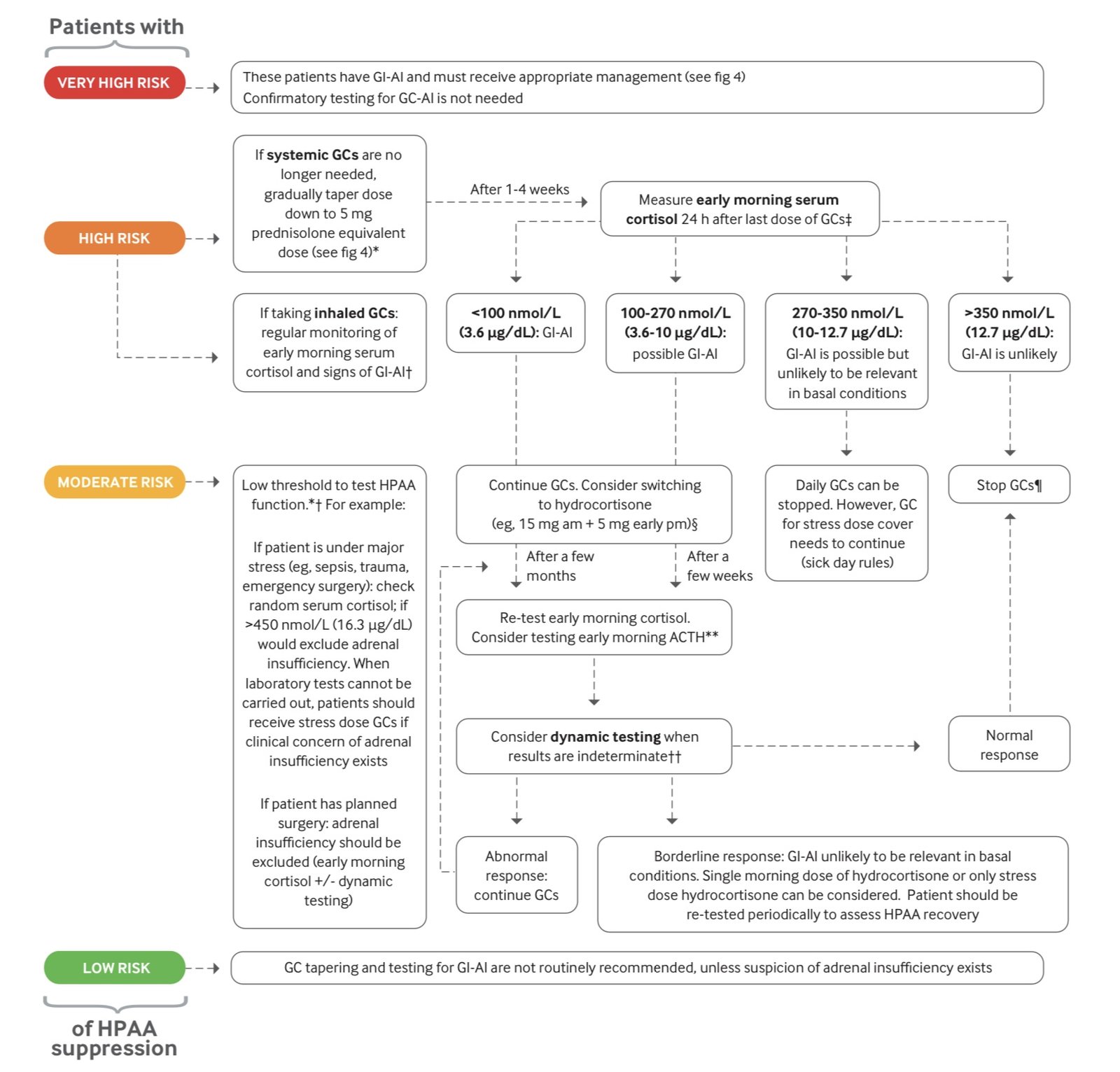
Source: BMJ 2021;374:n1380 | doi: 10.1136/bmj.n1380
Upcoming Incretin-Based Therapy on Phase 3 Clinical Trials for the Management of Diabetes and Obesity
Prof. Juan Pablo Frias
Orforglipron is an oral non-peptide GLP-1 receptor agonist with a recently published phase 2 trial for weight reduction in adults with obesity or overweight plus at least one weight-related health condition and without diabetes. Subjects were assigned to receive one of four doses of orforglipron (12mg, 24mg, 36mg or 45mg) or placebo for 36 weeks. Results showed that at week 26, patients receiving orfoglipron had lost approximately 9 to 13% of their body weight compared to 2% of patients receiving placebo. Gastrointestinal adverse events were similar to injectable GLP-1 RA but were most common mild to moderate in severity.
Retratrutide, an agonist of GIP, GLP1 and glucagon receptors (triagnoists), was also studied in adults without diabetes but with obesity or overweight plus > 1 weight relation condition. Subjects were assigned to receive 1mg, 2mg, 4mg, 8mg or 12mg for 48 weeks. Results showed a significant weight reduction across all doses of retratrutide compared to placebo. Gastrointestinal adverse events occurred with retratrutide and were more common at higher doses.